Improving Clinical Data Quality: From Manual Reporting to Intelligent Automation
Posted on: March 27th 2026
How can research teams ensure clinical trial data remains accurate and trustworthy as datasets continue to grow?
In clinical research, data quality reports play a critical role in assessing dataset accuracy, completeness, and reliability. However, as clinical trials generate large, multi-source datasets, traditional reporting approaches are becoming difficult to scale. Straive brings together AI-driven validation, data analytics, and operational workflows to streamline data quality evaluation and reporting.
Why Data Quality Is Critical in Clinical Research?
Clinical trials produce large, complex datasets needing continuous validation for accuracy and reliability. Poor data quality monitoring and reporting can lead to minor discrepancies that delay regulatory approval and impact patient safety and outcomes. Listing down key reasons why data quality is critical in clinical research:
Growing Volume and Complexity of Clinical Datasets
Clinical trials now gather data from diverse sources, electronic systems, labs, imaging, wearables, and patient reports. Validating and managing this multi-source data increases complexity for research teams.
Regulatory Scrutiny and Compliance Requirements
Regulators expect clinical data to be accurate, traceable, and audit-ready. The U.S. Food and Drug Administration (FDA) emphasizes that poor data integrity can undermine the ability to properly evaluate drug safety and effectiveness.
Impact of Poor Data Quality on Patient Safety and Outcomes
Clinical data plays a key role in decisions about the safety and effectiveness of treatments. When data is incomplete or inconsistent, researchers may misread results or overlook early signs of potential safety concerns.
The Cost of Inaccurate, Incomplete, or Inconsistent Data
Poor data quality also creates financial risk. According to Gartner, organizations lose an average of $12.9 million every year due to poor data quality.
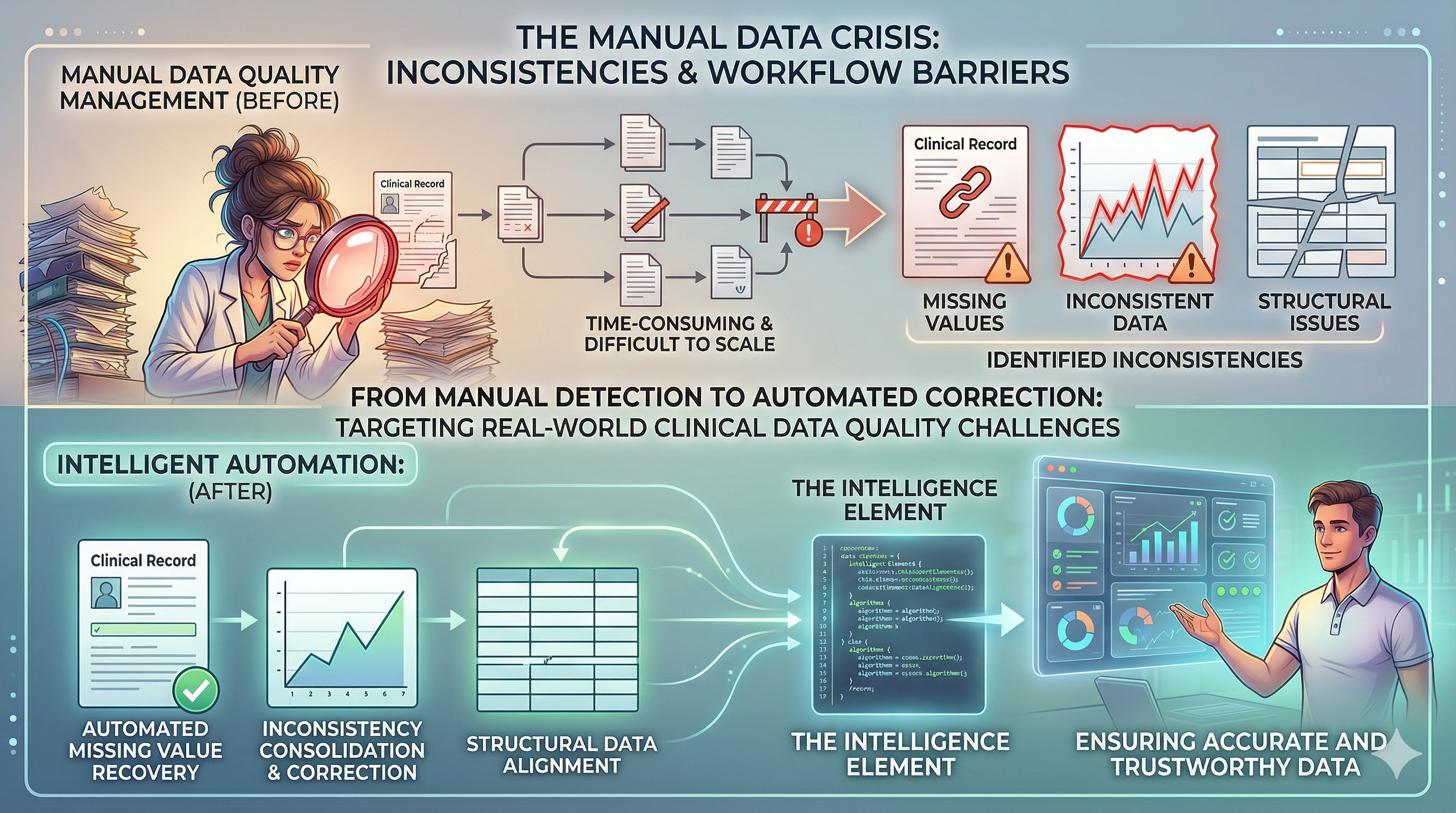
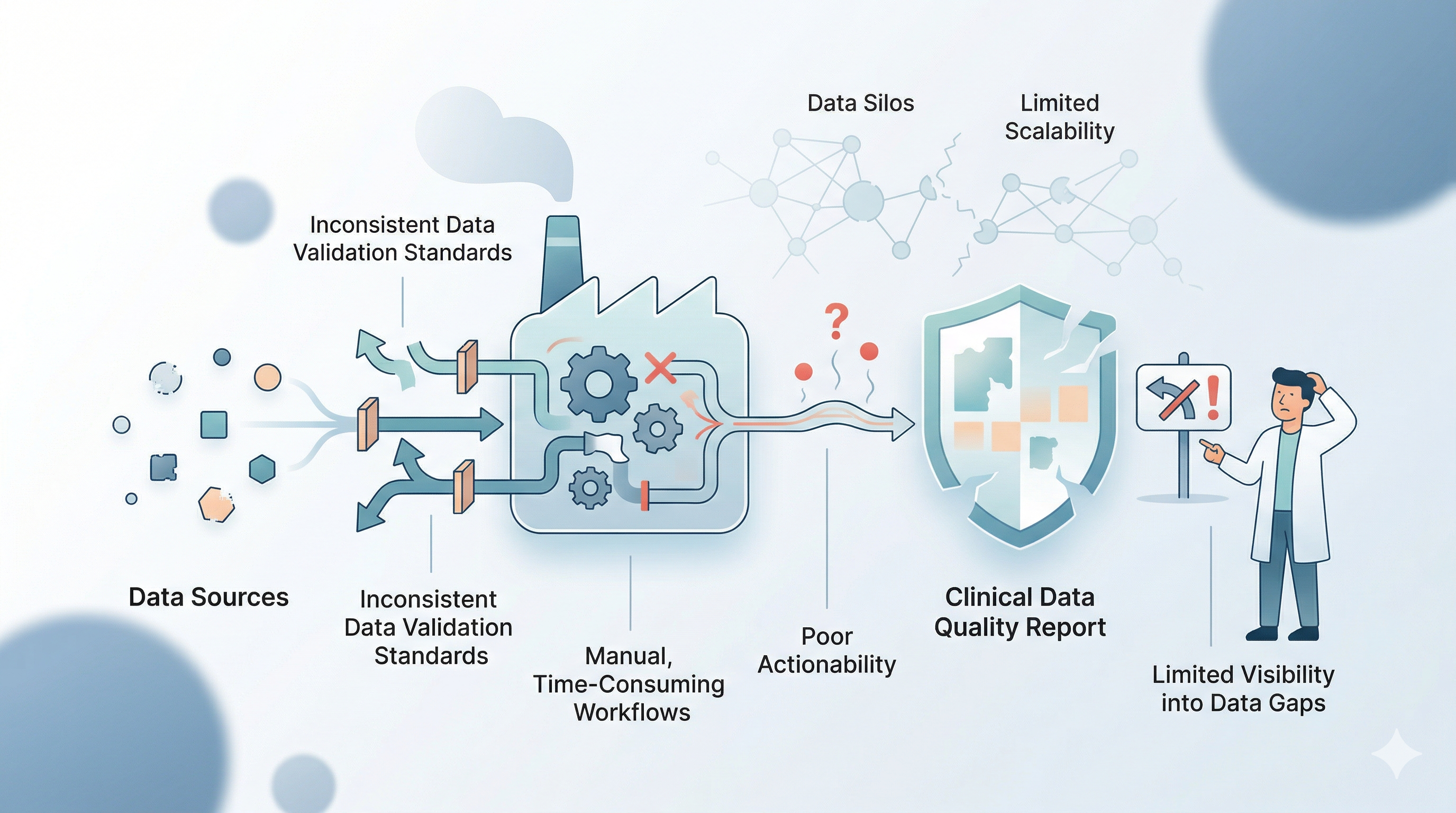
Common Challenges in Clinical Data Quality Reporting
Clinical teams rely on data quality reports to assess dataset accuracy and completeness. However, generating timely, consistent, and actionable reports remains a challenge as data volumes grow and sources become more fragmented.
- Manual and spreadsheet-based workflows delay report generation and lead to late identification of data issues
- Different teams and studies follow inconsistent validation approaches, resulting in uneven data quality standards
- Reports highlight issues but often lack clear visibility into root causes, making resolution difficult
- Outputs are not always actionable, leaving teams unclear on how to prioritize and fix issues
- Traditional reporting approaches struggle to scale with growing data volumes and complexity
- Data stored across multiple systems creates silos, making governance and consistency harder to maintain
Need for Automated, Intelligent Data Quality Reporting
Clinical teams use data quality reports to check whether trial datasets are accurate, complete, and ready for analysis or regulatory submission. When these reports take too long to generate or fail to highlight the right issues, valuable time is lost identifying and fixing data gaps.
Automation is changing how this process works. Automated systems can continuously review datasets and flag inconsistencies, missing values, and structural errors as new data is added.
The need for faster data quality evaluation is clear. Research from McKinsey shows that as much as 80 percent of the time in data workflows is spent cleaning and preparing data rather than analyzing it. For clinical teams working under tight timelines, reducing this effort is critical.
Automated reporting frameworks apply consistent validation rules and convert complex datasets into clear insights that teams can quickly review and act on.
How an AI-Powered Data Quality Reporting Solution Solves These Challenges
AI-powered reporting accelerates clinical dataset evaluation by consistently and rapidly identifying missing values, inconsistencies, and structural issues, moving beyond slow manual reviews.
At Straive, we help clinical teams simplify data quality evaluation by merging AI-driven validation with structured reporting. This allows researchers to quickly assess dataset health, find gaps, and correct issues earlier in the research process.
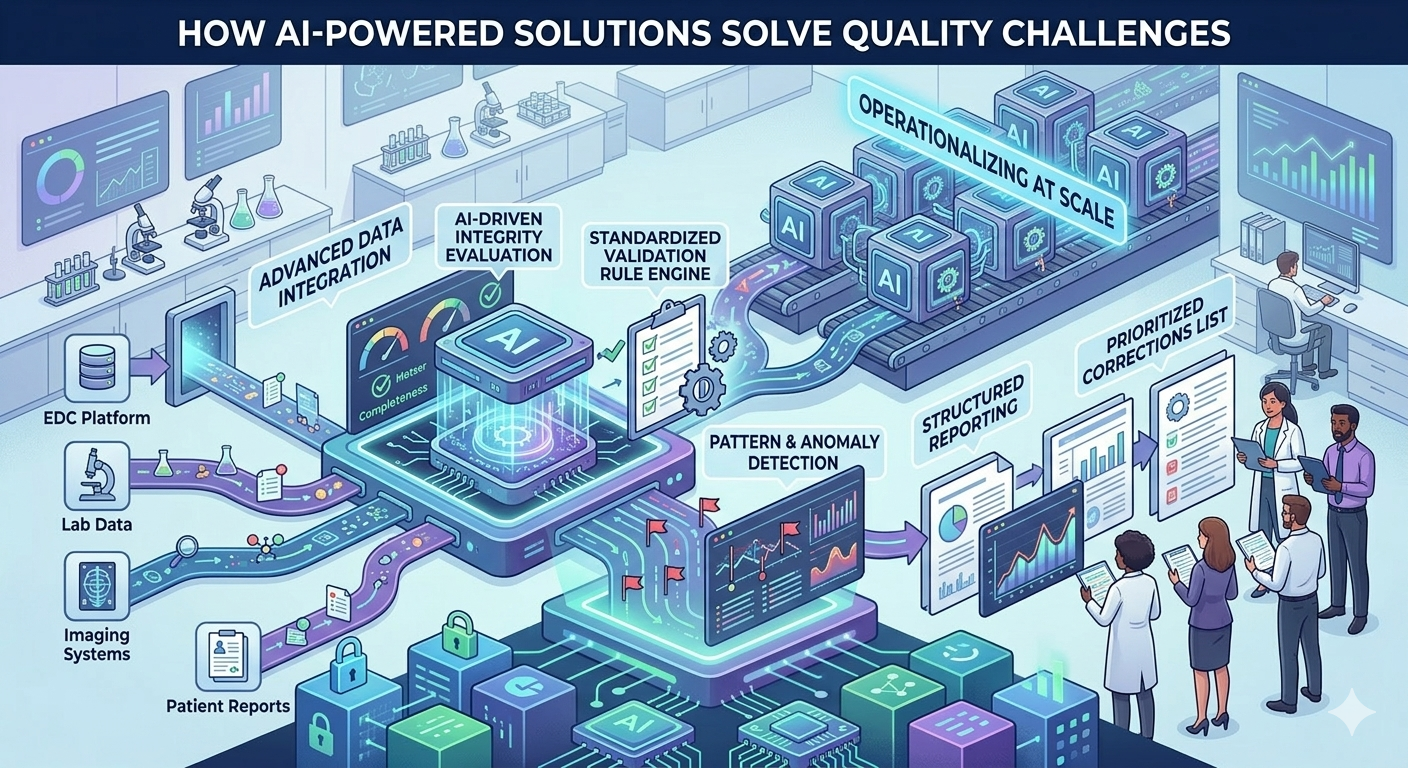
AI-Driven Data Integrity Evaluation
AI models continuously assess dataset accuracy, completeness, and consistency, helping teams identify potential data quality issues earlier. The system automates these checks across datasets, reducing manual effort and ensuring consistent evaluation without relying on individual team practices.
Advanced Data Integration Across Clinical Systems
Clinical data from EDC platforms, laboratories, imaging systems, and patient-reported sources can be unified and evaluated consistently. Data from multiple sources is brought into a unified view, eliminating the need for manual consolidation before quality assessment.
Standardized Data Quality Validation
AI-enabled validation frameworks apply consistent quality rules across datasets, helping maintain reliable data and support regulatory readiness. Validation rules are applied uniformly across studies, ensuring consistency and reducing dependency on manual or team-specific checks.
AI-Based Pattern and Anomaly Detection
Machine learning models identify unusual patterns, inconsistencies, and anomalies that traditional manual review processes often miss. This helps detect hidden data issues early, especially in large datasets where manual review is time-consuming and error-prone.
Data-Driven Quality Insights for Research Teams
Structured reporting converts complex clinical datasets into clear insights that help teams quickly identify gaps and prioritize corrections. It generates ready-to-use data quality reports that clearly highlight issues, gaps, and next steps, reducing the need for manual analysis and interpretation.
Operationalizing Data Quality Reporting at Scale
Automated reporting enables faster evaluation of clinical datasets, allowing research teams to focus on improving data quality and supporting better study outcomes. Reports are generated continuously as new data is added, enabling ongoing monitoring instead of delayed, periodic reviews.
Business Impact for Clinical and Research Teams
Stronger data quality reporting enables research teams to analyze results with more confidence and manage clinical studies more effectively. AI-enabled validation and structured reporting frameworks are helping organizations reduce operational risks, strengthen regulatory readiness, and accelerate the generation of research insights.
Stronger Regulatory Confidence And Audit Readiness
Validation methods ensure that data remains trustworthy for regulators, helping clinical submissions remain transparent during audits.
Reduced Risk Of Costly Data Errors
Data checks help identify issues early in the data process, minimizing the likelihood of costly errors during analysis or regulatory submissions.
Faster Insights From Complex Clinical Data
AI-enabled analytics helps research teams process large and diverse clinical datasets more efficiently, enabling faster interpretation of trial data and operational insights.
Improved Biomedical Data Curation And Structuring
AI-driven biomedical data curation has demonstrated over 90% accuracy in entity annotation while automating more than 80% of curation workflows, improving how large scientific datasets are prepared for research analysis. Read
Enhanced Safety Monitoring Through AI Analytics
Advanced analytics and AI-driven monitoring frameworks are helping pharmacovigilance teams analyze large volumes of safety data more efficiently, improving the detection of potential safety signals and strengthening regulatory reporting processes.
The Future of Clinical Data Quality Is Intelligent and Automated
Clinical research is rapidly moving toward a future in which data quality must be continuously evaluated rather than reviewed after problems arise. As clinical datasets grow in scale and complexity, research teams need smarter ways to assess data integrity and maintain confidence in trial results.
The next phase of clinical data management will depend on intelligent systems that can automatically evaluate datasets, surface quality gaps, and convert complex clinical data into clear, actionable insights.
By enabling faster data integrity evaluation and structured reporting, AI-driven approaches allow research teams to identify issues earlier, strengthen dataset reliability, and support more confident clinical and regulatory decisions.
See It in Action
See how intelligent automation can simplify clinical data quality reporting and help teams identify inconsistencies faster. Explore how AI-driven validation and analytics transform complex datasets into clear, actionable insights for research teams.
Request a demo to see how AI-driven reporting can transform clinical data quality management.

Santosh Shevade is a Principal Data Consultant at Gramener – A Straive Company. With deep expertise in healthcare strategy, digital health, and clinical development and operations, he has supported nearly 50 clinical development programs across all clinical phases. His experience spans advanced analytics solution design for pharmaceutical companies, mHealth implementation, and AI applications in healthcare. Previously at Novartis and Johnson & Johnson, Santosh led global clinical development teams and streamlined data review processes for major regulatory submissions. A certified MBTI trainer and leadership coach, he serves as visiting faculty at ISB Hyderabad and Welingkar Institute, focusing on healthcare technology innovation and biopharma strategy.