Why Fragmented Content Ecosystems Are Slowing Pharma Operations
Posted on: March 26th 2026
Pharma as a Content-Critical Enterprise
Pharmaceutical organizations operate in an environment where content is inseparable from data. Every stage of the product lifecycle generates critical documentation, including clinical study reports and pharmacovigilance updates, regulatory submissions, labeling components, investigator brochures, and patient-facing materials. Each document directly supports product approval, market access, and overall pharma regulatory compliance.
Regulatory expectations continue to intensify. Agencies such as the FDA, EMA, and ICH require transparency, traceability, and demonstrable data integrity across submissions. It is no longer sufficient to submit accurate documents. Organizations must demonstrate how information evolved, who approved specific changes, and how alignment is maintained across regions and versions. Pharma regulatory compliance now depends as much on content governance as it does on scientific evidence.
In this context, content functions as operational infrastructure. It underpins regulatory content management, scientific credibility, and global coordination. However, many pharmaceutical document management environments have evolved function by function rather than as part of a unified design. As pharma digital transformation accelerates, this structural gap becomes increasingly visible.
Pharma is a content-driven enterprise Regulatory submissions, clinical reports, labeling, and safety documentation make content a core operational asset across the pharmaceutical lifecycle. |
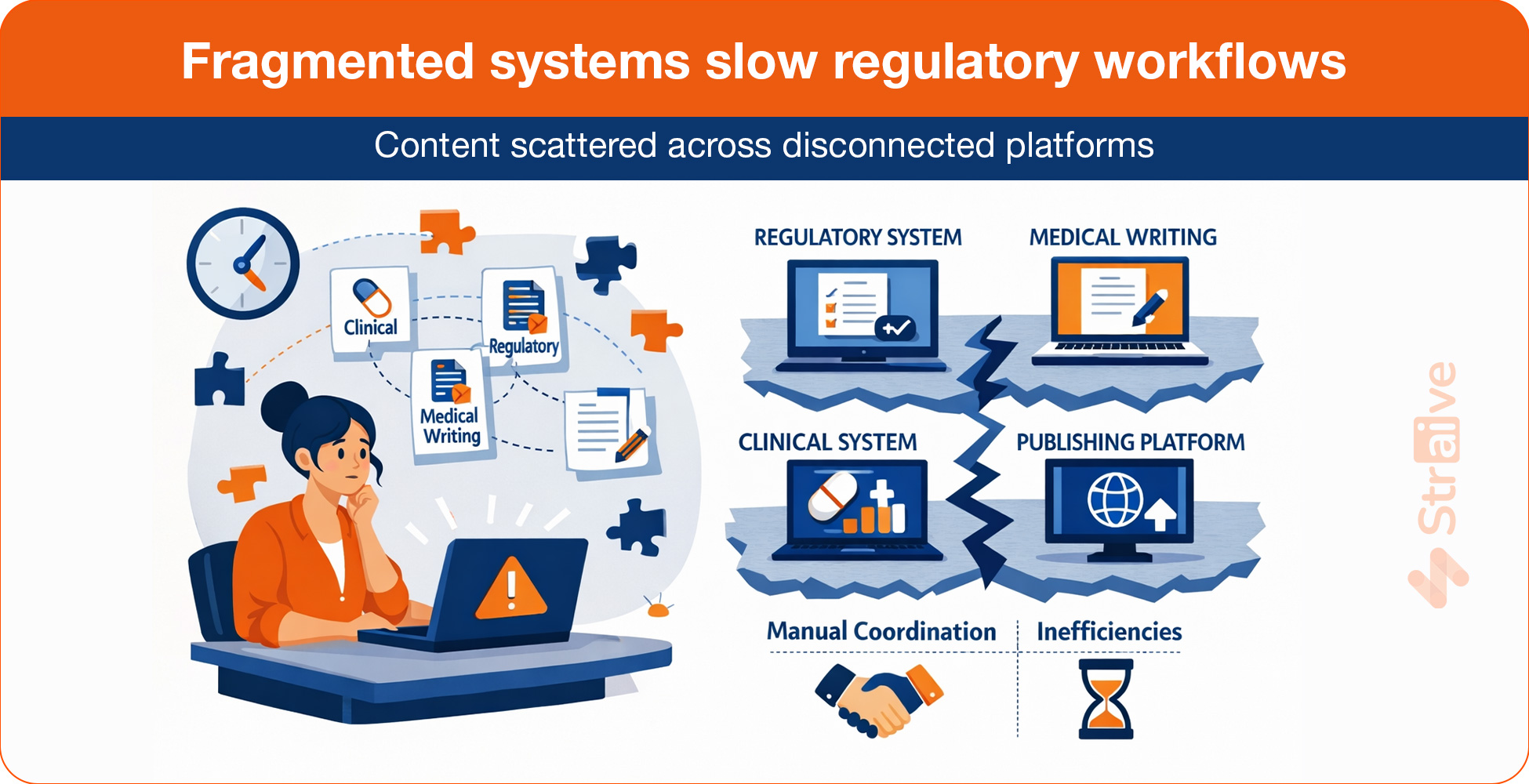
The Fragmentation Challenge
In many pharmaceutical organizations, content does not reside within a single, unified ecosystem. Regulatory Affairs may operate within one repository. Clinical Operations and Medical Writing often rely on separate authoring platforms. Publishing teams manage formatting and submission tools independently. Regional affiliates maintain localized document versions. Safety data may be stored in additional systems.
Each system serves a defined purpose. Collectively, however, they do not always operate as a coordinated regulatory content management framework.
Workflows frequently depend on manual coordination. Teams exchange documents through email. Files are compared across versions. Structured data and narrative content are often managed separately, without shared metadata standards or integrated governance. Limited interoperability between systems means updates must be manually transferred from one environment to another.
Fragmented systems slow regulatory workflows Content often sits across disconnected platforms used by regulatory, clinical, medical writing, and publishing teams, creating manual coordination and inefficiencies. |
As portfolios expand and global launches occur in parallel rather than sequence, fragmented pharmaceutical document management models become increasingly difficult to sustain.
Manual Dependencies and Version Drift
The operational consequences of fragmentation are most visible during routine updates.
Consider a safety revision. A single change may need to be reflected across regulatory submissions, labeling documentation, patient information leaflets, and internal reference materials. In a disconnected ecosystem, each document must be revised and reviewed separately. There is no centralized control layer ensuring that updates cascade consistently across assets.
Human-dependent tagging and formatting introduce further variability. Teams may interpret language differently. Regional affiliates may adjust phrasing to align with local regulatory expectations. Departments may independently update overlapping materials without shared oversight.
Over time, these dependencies create version drift. Statements that were once aligned begin to vary subtly across documents and regions. Reconciling differences requires additional review cycles. Regulatory and scientific teams devote time to validating consistency instead of advancing substantive improvements.
Version drift creates compliance risk When multiple teams update documents across systems without centralized governance, inconsistencies emerge that require repeated reconciliation cycles. |
Minor operational friction gradually accumulates, slowing momentum across the broader pharma digital transformation journey.
Regulatory and Operational Exposure
Fragmentation does more than slow processes; it increases exposure.
Regulators expect organizations to maintain clear audit trails and demonstrable data lineage. During inspections, companies must show how content evolved, who approved changes, and how consistency was preserved across markets and submissions. Effective pharma regulatory compliance depends on reliable traceability.
In disconnected systems, this information may exist but be distributed across platforms. Version histories are not always consolidated. Demonstrating alignment across global submissions can require manual reconstruction. Inspection readiness becomes an exercise in assembling evidence rather than relying on built-in controls.
Repeated review cycles and late-stage discrepancies increase the risk of submission delays and regulatory scrutiny. What should be embedded within pharmaceutical document management systems instead becomes reactive and resource-intensive.
These systemic challenges are examined in greater depth in our white paper, which explores how fragmented content ecosystems affect regulatory timelines, compliance confidence, and global collaboration among pharmaceutical organizations.
Business Impact of Fragmentation
Operational inefficiencies inevitably translate into measurable business consequences.
Extended review cycles can delay regulatory submissions and affect time-to-market. Rework increases cost. Highly trained regulatory and scientific professionals allocate time to reconciling discrepancies rather than focusing on strategy and innovation.
As organizations manage multiple products across multiple regions, reduced agility becomes more pronounced. Coordinating updates across geographies demands additional oversight. Simultaneous lifecycle changes increase complexity within regulatory content management processes. Content that should support development and commercialization becomes a constraint.
Under intensifying regulatory scrutiny and expanding global scale, the cumulative cost of fragmentation becomes difficult to ignore.
Why Incremental Fixes Are Insufficient
Organizations often respond with incremental improvements. Additional reviewers are introduced to strengthen oversight. Point solutions automate isolated tasks. Manual governance processes are reinforced.
While these measures may improve specific activities, they do not resolve structural disconnects. Adding reviewers does not eliminate version drift when systems remain siloed. Isolated automation cannot overcome limited interoperability between core pharmaceutical document management platforms. Manual controls do not scale effectively as submission volumes grow and global regulatory demands increase.
Fragmentation reflects architectural limitations, not simply process inefficiency. Addressing it requires structural alignment rather than incremental adjustment.
Transition to a Structural Solution
Moving beyond fragmentation requires a shift toward an integrated structural foundation.
A unified, scalable content backbone establishes a centralized system of record across regulatory, clinical, medical writing, and publishing functions. Platforms exchange information within defined governance boundaries. Regulatory content management becomes coordinated rather than compartmentalized.
Pharma needs a unified content backbone Integrated regulatory content management systems enable traceability, governance, and scalable collaboration across global teams. |
Compliance by design embeds governance directly into workflows. Metadata-driven controls reduce duplication and enable consistent reuse. Audit trails and data lineage become inherent system capabilities rather than information reconstructed during inspections.
Such structural alignment strengthens pharma regulatory compliance while supporting broader pharma digital transformation objectives.
The whitepaper outlines this scalable backbone model in detail, describing its core capabilities and a phased implementation roadmap aligned with global regulatory expectations.
From Fragmentation to a Scalable Content Backbone
Fragmented content ecosystems are increasingly unsustainable in a landscape defined by regulatory scrutiny, global expansion, and digital transformation. What once appeared manageable now introduces delays, reconciliation efforts, and compliance exposure.
Pharma leaders must evaluate whether their current regulatory content management and pharmaceutical document management systems support innovation and scalability, or whether structural constraints are limiting progress.
For a comprehensive roadmap to transforming fragmented content operations into a unified and scalable backbone, read the whitepaper.

Straive helps clients operationalize the data> insights> knowledge> AI value chain. Straive’s clients extend across Financial & Information Services, Insurance, Healthcare & Life Sciences, Scientific Research, EdTech, and Logistics.